How to Calculate Moles: A Comprehensive Guide
Understanding moles is fundamental to chemistry. A mole isn't a furry creature; it's a unit representing a specific number of particles (atoms, molecules, ions, etc.). This guide will walk you through how to calculate moles using different methods, ensuring you master this crucial concept.
What is a Mole?
Before diving into calculations, let's solidify the definition. A mole (mol) is the amount of substance containing Avogadro's number (approximately 6.022 x 10²³) of elementary entities. This number is incredibly large, reflecting the tiny size of atoms and molecules. Think of it like a dozen (12) but on a much grander scale for the chemical world.
Calculating Moles: Key Formulas
Several formulas help calculate moles, depending on the information available. Here are the most common ones:
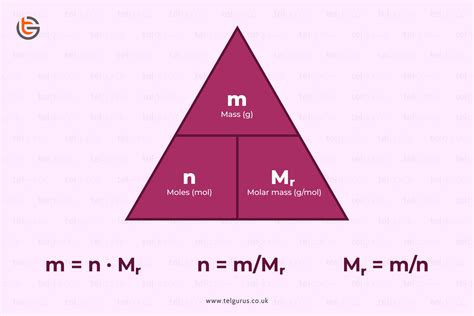
1. Using Mass and Molar Mass
This is the most frequently used method. The formula is:
Moles (mol) = Mass (g) / Molar Mass (g/mol)
- Mass (g): The mass of the substance in grams. You'll need a balance or scale to measure this.
- Molar Mass (g/mol): The mass of one mole of the substance. You can find this on the periodic table for elements or calculate it by adding the atomic masses of all atoms in a molecule.
Example: Calculate the number of moles in 10 grams of water (H₂O).
The molar mass of water is: (2 x 1.01 g/mol for H) + (1 x 16.00 g/mol for O) = 18.02 g/mol
Moles = 10 g / 18.02 g/mol ≈ 0.555 mol
2. Using Number of Particles and Avogadro's Number
This method is useful when dealing with the actual number of atoms, molecules, or ions. The formula is:
Moles (mol) = Number of Particles / Avogadro's Number (6.022 x 10²³)
Example: Calculate the number of moles in 3.011 x 10²³ atoms of carbon.
Moles = 3.011 x 10²³ / 6.022 x 10²³ ≈ 0.5 mol
3. Using Molarity and Volume (for solutions)
When working with solutions, molarity (moles per liter) is often used. The formula is:
Moles (mol) = Molarity (mol/L) x Volume (L)
Example: Calculate the number of moles in 250 mL of a 0.1 M solution of sodium chloride (NaCl).
First, convert mL to L: 250 mL = 0.25 L
Moles = 0.1 mol/L x 0.25 L = 0.025 mol
Tips for Accurate Mole Calculations
- Unit consistency: Ensure all your units are consistent (grams for mass, liters for volume).
- Significant figures: Pay attention to significant figures in your calculations. Your answer should reflect the precision of your measurements.
- Periodic table: Always have a periodic table handy to look up atomic masses.
- Practice: The best way to master mole calculations is through practice. Work through numerous examples to build your confidence.
Mastering Mole Calculations: Beyond the Basics
This guide provides the foundational knowledge for calculating moles. Further exploration into stoichiometry, which uses mole calculations to determine reactant and product quantities in chemical reactions, will solidify your understanding. With consistent practice and a solid grasp of these fundamental formulas, you'll confidently navigate the world of mole calculations.